Growth factor refers to a class of biologically active polypeptides that can promote cell growth and development. They regulate the proliferation and differentiation of various cells through autocrine and or paracrine ways. Like other cytokines, growth factors' action is through binding to specific cell membrane receptors with high affinity, that is, growth factor receptors. There are many kinds of growth factors, such as epidermal growth factor (EGF), insulin (Insulin), platelet-derived growth factor (PDGF), vascular endothelial growth factor (VEGF), fibroblast growth factor (FGF), glial neurotrophic factor (GDNF) etc. The specific receptors for different growth factors are different, but they are all transmembrane proteins, and most of them have tyrosine kinase activity (Figure 1).
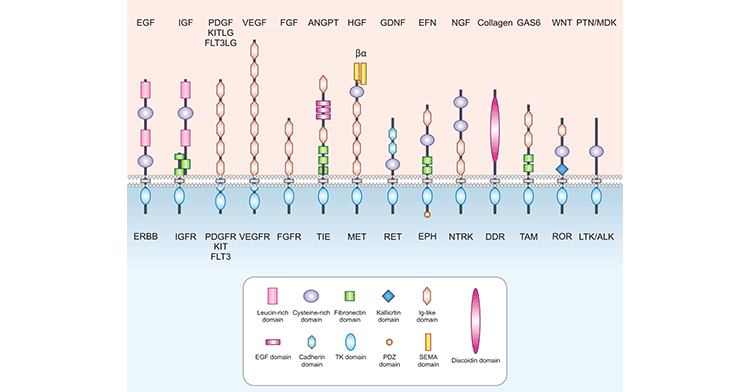
Figure 1. The structures of common growth factor receptors
The following takes the common epidermal growth factor receptor (EGFR) and insulin receptor (INSR) as examples to illustrate the structure and function of growth factor receptors.
Epidermal growth factor receptor (EGFR) is a cell surface receptor for the extracellular protein ligand of epidermal growth factor (EGF). EGFR is one of four members of the ErbB receptor family, also known as HER1 or ErbB1. EGFR is a transmembrane receptor with tyrosine kinase activity and belongs to the receptor tyrosine kinase family (RTKs). It is activated by binding to ligands, including EGF and TGFα. After activation, EGFR is converted from monomer to dimer. The dimerization of EGFR involves both the binding of two cognate receptors (homodimerization) and the binding of different members of the human EGF-related receptor (HER) tyrosine kinase family (hetero-dimerization).
After dimerization, EGFR can phosphorylate and activate its intracellular tyrosine residues, including activation sites such as Y992, Y1045, Y1068, Y1148, and Y1173. This autophosphorylation can initiate intracellular signal transduction (including MAPK, Akt and JNK pathways), regulate the transcription of genes activated by transcription factors through the cascade reaction of adaptor proteins and enzymes in the cytoplasm, and then guide cell migration, adhesion, proliferation, and differentiation. , apoptosis.
The insulin receptor (INSR), as its name implies, is a receptor that specifically binds to insulin and belongs to the Ò-type subfamily of RTKs. INSR is a tetramer consisting of two alpha subunits and two beta subunits connected by disulfide bonds. The two α subunits are located on the outside of the cytoplasmic membrane, where there are binding sites for insulin; the two β subunits are transmembrane proteins that play a role in signal transduction. The classical signaling pathway of INSR is triggered by binding to insulin. After insulin binds to INSR, INSR undergoes autophosphorylation and its tyrosine kinase activity is activated. The latter are able to recruit and phosphorylate various substrate docking proteins, such as the insulin receptor substrate (IRS) family of proteins. Phosphorylated IRS exposes binding sites for numerous signaling partners to bind, including the PI3K-AKT signaling pathway. The PI3K-AKT signaling pathway regulates cellular physiological activities such as glucose transport, glycogen synthesis, and protein translation by activating multiple downstream target proteins.
Regard reading you may be interested in...
The overview of Wnt signaling pathway
CUSABIO has listed some popular targets related growth factor receptor. Click on the corresponding target to view all the reagents related to the target.
Receptor by Differentiation Cell
Receptor by Drug Target
Related Articles