Key Points
-
Implantable cardioverter-defibrillators (ICDs) reduce mortality in patients with increased risk of sudden cardiac death, but use of these devices has been limited by complications with the intravascular leads
-
The subcutaneous ICD (S-ICD) is a new ICD device that avoids placing leads in the cardiac vascular system
-
The S-ICD is safe and effective for the treatment of ventricular tachycarrhythmias, but use of S-ICDs is limited by the lack of pacing or remote monitoring capabilities
-
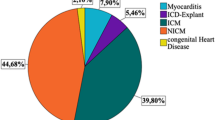
The S-ICD is most commonly used in young patients at risk for ventricular fibrillation, but not ventricular tachycardia, and without pacing indications
-
Future generations of the S-ICD should allow for expanded use and for subcutaneous leads to be combined with leadless pacing, enabling the S-ICD to become the standard ICD device
Abstract
The subcutaneous implantable cardioverter-defibrillator (S-ICD) is a novel technology for the treatment of sudden cardiac death. The system consists of a pulse generator implanted in the left axillary position and a single subcutaneous lead for detection and delivery of therapy. Initial clinical trials of S-ICDs demonstrated improved safety and efficacy when compared to transvenous ICD systems, leading to their widespread approval. The main advantage of the S-ICD is the avoidance of vascular access and the complications associated with transvenous leads. Owing to limitations of S-ICDs, patients who require pacing support or antitachycardia pacing are not candidates for the device; instead, this system is currently used most commonly in young patients with previous lead malfunction, limited vascular access, or low risk for subsequent bradycardia or antitachycardia pacing. Findings from device trials support S-ICDs as a viable alternative to transvenous ICDs in certain patients, and the current limitations associated with S-ICDs are likely to be addressed in future iterations of the device, extending its indications and target patient populations.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Zipes, D. P. & Wellens, H. J. Sudden cardiac death. Circulation 90, 2334–2351 (1998).
Rajamani, K., Goldberg, A. S. & Wilkoff, B. L. Shock avoidance and the newer tachycardia therapy algorithms. Cardiol. Clin. 32, 191–200 (2014).
van Rees, J. B. et al. Implantation-related complications of implantable cardioverter-defibrillators and cardiac resynchronization therapy devices: a systematic review of randomized clinical trials. J. Am. Coll. Cardiol. 58, 995–1000 (2011).
Stephenson, E. A. et al. A multicenter experience with novel implantable cardioverter defibrillator configurations in the pediatric and congenital heart disease population. J. Cardiovasc. Electrophysiol. 17, 41–46 (2006).
Jaroszewski, D. E. et al. Nontraditional surgical approaches for implantation of pacemaker and cardioverter defibrillator systems in patients with limited venous access. Ann. Thorac. Surg. 88, 112–116 (2009).
Bardy, G. H. et al. An entirely subcutaneous implantable cardioverter-defibrillator. N. Engl. J. Med. 363, 36–44 (2010).
Rowley, C. P. & Gold, M. R. Subcutaneous implantable cardioverter defibrillator. Circ. Arrhythm. Electrophysiol. 5, 587–593 (2012).
Gold, M. R. et al. Head-to-head comparison of arrhythmia discrimination performance of subcutaneous and transvenous ICD arrhythmia detection algorithms: the START study. J. Cardiovasc. Electrophysiol. 23, 359–366 (2012).
Weiss, R. et al. Safety and efficacy of a totally subcutaneous implantable cardioverter-defibrillator. Circulation 128, 944–953 (2013).
Boston Scientific. S-ICD frequently asked questions [online]. (2014).
Killingsworth, C. R., Melnick, S. B., Litovsky, S. H., Ideker, R. E. & Walcott, G. P. Evaluation of acute cardiac and chest wall damage after shocks with a subcutaneous implantable cardioverter-defibrillator in swine. Pacing Clin. Electrophsiol. 36, 1265–1272 (2013).
Brady, P. A. et al. High failure rate for an epicardial implantable cardioverter-defibrillator lead: implications for long-term follow-up of patients with an implantable cardioverter-defibrillator. J. Am. Coll. Cardiol. 31, 616–622 (1998).
Jarman, J. W. & Todd, D. M. United Kingdom national experience of entirely subcutaneous implantable cardioverter-defibrillator technology: important lessons to learn. Europace 15, 1158–1165 (2013).
Kleeman, T. et al. Annual rate of transvenous defibrillation lead defects in implantable cardioverter-defibrillators over a period of >10 years. Circulation 115, 2474–2780 (2007).
Abkenari, L. D. et al. Clinical experience with a novel subcutaneous implantable defibrillator in a single center. Clin. Res. Cardiol. 100, 737–744 (2011).
Jarman, J. W. E. et al. Clinical experience of entirely subcutnaneous implantable cardioverter-defibrillators in children and adults: cause for caution. Eur. Heart J. 33, 1351–1359 (2012).
Aydin, A. et al. Shock efficacy of subcutaneous implantable caridoverter-defibrillators for prevention of sudden cardiac death. Circ. Arrhythm. Electrophysiol. 5, 913–919 (2012).
Nordkamp, L. et al. The entirely subcutaneous implantable cardioverter-defibrillator: initial clinical experience in a large Dutch cohort. J. Am. Coll. Cardiol. 60, 1933–1939 (2012).
Kobe, J. et al. Implantation and follow-up of totally subcutaneous versus conventional implantable cardioverter-defibrillators: a multicenter case-control study. Heart Rhythm 10, 29–36 (2013).
Pedersen, S. S. et al. Evaluation of factors impacting clinical outcome and cost effectiveness of the S-ICD: design and rationale of the EFFORTLESS S-ICD Registry. PACE 35, 574–579 (2012).
Lambiase, P. D. et al. Worldwide experience with a totally subcutaneous implantable defibrillator: early results from the EFFORTLESS S-ICD registry. Eur. Heart J. 35, 1657–1665 (2014).
Hauser, R. G. et al. Longevity of sprint fidelis implantable cardioverter-defibrillator leads and risk factors for failure. Circulation 123, 358–363 (2011).
Moss, A. J. et al. Reduction in inappropriate therapy and mortality through ICD programming. N. Engl. J. Med. 367, 2275–2283 (2012).
Gold, M. R. et al. The use of discrimination algorithm to reduce inappropriate shocks with a subcutaneous ICD. Heart Rhythm 11, 1352–1358 (2014).
Groh, C. A. et al. Use of an electrocardiographic screening tool to determine candidacy for a subcutaneous implantable cardioverter-defibrillator. Heart Rhythm 11, 1361–1366 (2014).
Kooiman, K. M. et al. Inappropriate subcutaneous implantable cardioverter-defibrillator shocks due to T-wave oversensing can be prevented: implications for management. Heart Rhythm 11, 426–434 (2014).
Lupo, P. P., Pelissero, G., Ali, H., Sanghera, R. & Cappato, R. Development of an entirely subcutaneous implantable cardioverter-defibrillator. N. Engl. J. Med. 363, 36–44 (2010).
Aziz, S., Leon, A. R. & El-Chami, M. F. The subcutaneous defibrillator. J. Am. Coll. Cardiol. 63, 1473–1479 (2014).
Saxon, L. The subcutaneous implantable defibrillator: a new technology that raises an existential question for the implantable cardioverter-defibrillator. Circulation 128, 938–940 (2013).
Thijssen, J. et al. Implantable cardioverter-defibrillator longevity under clinical circumstances: an analysis according to device type, generation, and manufacturer. Heart Rhythm 9, 513–519 (2012).
Hauser, R. G. The subcutaneous implantable cardioverter-defibrillator: should patients want one? J. Am. Coll. Cardiol. 61, 20–22 (2013).
Bardy, G. H. et al. Amiodarone or an implantable cardioverter-defibrillator for congestive heart failure. N. Engl. J. Med. 352, 225–237 (2005).
Moss, A. J. et al. Prophylactic implantation of a defibrillator in patients with myocardial infarction and reduced ejection fraction. N. Engl. J. Med. 346, 877–883 (2002).
Kloppe, A. et al. Effect of long detection interval implantable cardioverter-defibrillator settings in secondary prevention poplulation: data from the ADVANCE III trial. Circulation 130, 308–314 (2014).
Poole, J. E. & Gold, M. R. Who should receive the subcutaneous implantable cardioverter-defibrillator? The subcutaneous implantable cardioverter-defibrillator should be considered in all patients who do not require pacing. Circ. Arrhythm. Electrophysiol. 6, 1236–1244 (2013).
Guedon-Moreau, L. et al. A randomized study of remote follow-up of implantable cardioverter-defibrillators: safety and efficacy report of the ECOST trial. Eur. Heart J. 34, 605–614 (2013).
Saxon, L. A. et al. Long-term outcome after ICD and CRT implantation and influence of remote device follow-up: the ALTITUDE survival study. Circulation 122, 2359–2367 (2010).
Author information
Authors and Affiliations
Contributions
Both authors researched data for the article, discussed its content, and wrote, reviewed, and edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
M.R.G. has received consulting fees and performed clinical trials with Boston Scientific, Medtronic and St Jude. G.F.L. declares no competing interests
Rights and permissions
About this article
Cite this article
Lewis, G., Gold, M. Clinical experience with subcutaneous implantable cardioverter-defibrillators. Nat Rev Cardiol 12, 398–405 (2015). https://doi.org/10.1038/nrcardio.2015.56
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrcardio.2015.56
This article is cited by
-
Comparing the safety of subcutaneous versus transvenous ICDs: a meta-analysis
Journal of Interventional Cardiac Electrophysiology (2021)
-
Heart failure with reduced ejection fraction
Nature Reviews Disease Primers (2017)